Treatments for Tuberculosis and Non‐tuberculous Mycobacterial Lung Infections: Dual‐targeted Rifamycin‐AAP Conjugates
Inventor(s): Richard H. Ebright
Awarded: October 2020
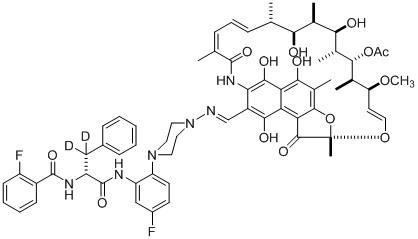
Summary: The team is developing a novel class of dual-targeted rifampin-AAP conjugates, "RifaAAPs," comprising the core pharmacophore of rifampin (Rif; first-line anti-TB drug that binds to the mycobacterial RNA polymerase active center and sterically blocks RNA synthesis) covalently linked to an Nα-aroyl-N-aryl-phenylalaninamide (AAP; synthetic anti-TB agent that binds to a different site on mycobacterial RNA polymerase and allosterically blocks RNA synthesis).
RifaAAPs inhibit mycobacterial RNA polymerase through two different binding sites (the Rif and AAP binding sites) and two different mechanisms (the Rif and AAP mechanisms). As a result, RifaAAPs are able to overcome Rif‐resistance and are able to overcome to AAP‐resistance. As a further result, RifaAAPs exhibit much lower‐‐orders of magnitude lower‐‐resistance emergence than Rif and AAP (with RifaAAP-resistance requiring a rare double mutational hit that alters both the Rif site and the AAP site, in contrast to Rif-resistance and AAP‐resistance, each of which requires only a single mutational hit that alters the Rif site or the AAP site).
RifaAAPs will provide:
- a shorter treatment duration and a higher treatment success rate for treatment of drug‐susceptible, drug-resistant, multi-drug-resistant, and extensively-drug-resistant TB, and
- a shorter treatment duration and a higher treatment success rate for treatment of nontuberculous mycobacterial lung infections.
Market Applications:
Target market and opportunity, TB:
- Addressable global market for TB is 10 million patients annually (580,000 DR, MDR, or XDR).
- Addressable US market for TB is 9,000 patients annually (100 DR, MDR, or XDR).
- Incidences are projected to be roughly level.
Target market and opportunity, NTM lung infections:
- Addressable global market for NTM lung infections is 2-4 million patients annually.
- Addressable US market for NTM lung infections is 180,000 patients annually.
- Incidences are projected to increase over the next decade.