- For Faculty & Staff
- Internal Research Funding Opportunities
- HealthAdvance
-
Funded Projects

HealthAdvance Funded Projects
Explore the latest projects funded through our funding platform that aims to speed up the translation of biomedical discoveries into commercially viable diagnostics, devices, therapeutics and tools to improve health and patient care.
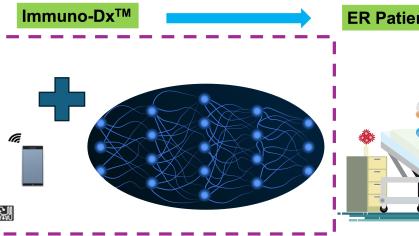
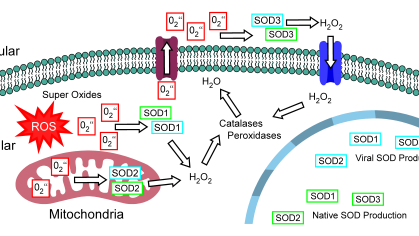
This project will develop a biomedical technology called Immuno-Dx, whose value proposition lies in its ability to provide vital information about patients’ immune response to infections, and to fulfill unmet need for personalized diagnostics of septic patients in emergency department (ED) settings.
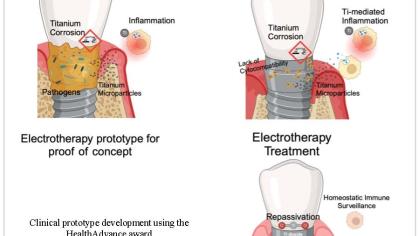
Our project aims to revolutionize peri-implantitis treatment by developing an innovative electrotherapy device designed to restore the passivation layer of titanium implants. By targeting the root cause of implant degradation and associated inflammation, we seek to enhance the longevity and success of dental implants and improve patient outcomes.
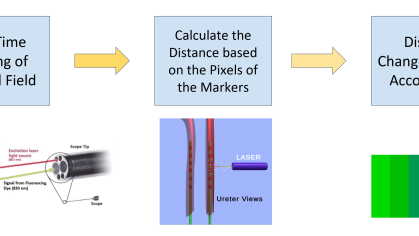
Rutgers researchers have developed an AI-based system to prevent iatrogenic ureteral injury in gynecologic and colorectal surgeries. The technology continuously tracks surgical instruments, providing real-time alerts or movement halts near the ureter. Platform-independent, it enhances intraoperative safety and surgeon control, reducing complications in complex cases with distorted anatomy.